Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|
|



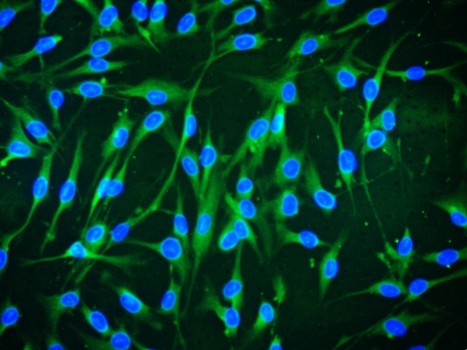



In an attempt to modulate mesenchymal stem cells (MSCs) allograft rejection in vivo, the authors transduced MSCs with an interleukin-13 (IL13)-expressing lentiviral vector. Their data clearly indicate that prolonged survival of IL13-expressing allogeneic MSC grafts in muscle tissue coincided with the induction of an alternatively activated macrophage phenotype in vivo and a reduced number of alloantigen-reactive IFNγ- and/or IL2-producing CD8+ T cells compared to nonmodified allografts. Similarly, intracerebral IL13-expressing MSC allografts also exhibited prolonged survival and induction of an alternatively activated macrophage phenotype, although a peripheral T cell component was absent. In summary, this study demonstrates that both innate and adaptive immune responses are effectively modulated in vivo by locally secreted IL13, ultimately resulting in prolonged MSC allograft survival in both muscle and brain tissue.